
Publication
Motor Finance Redress: The Way Ahead
On August 1, 2025, the UK Supreme Court delivered its long-awaited judgment in Hopcraft v Close Brothers Limited and on 3 August the FCA announced it would consult on a redress scheme.

Australia | Publication | September 2021
This article has been co-authored with Gianluca Tripodi.
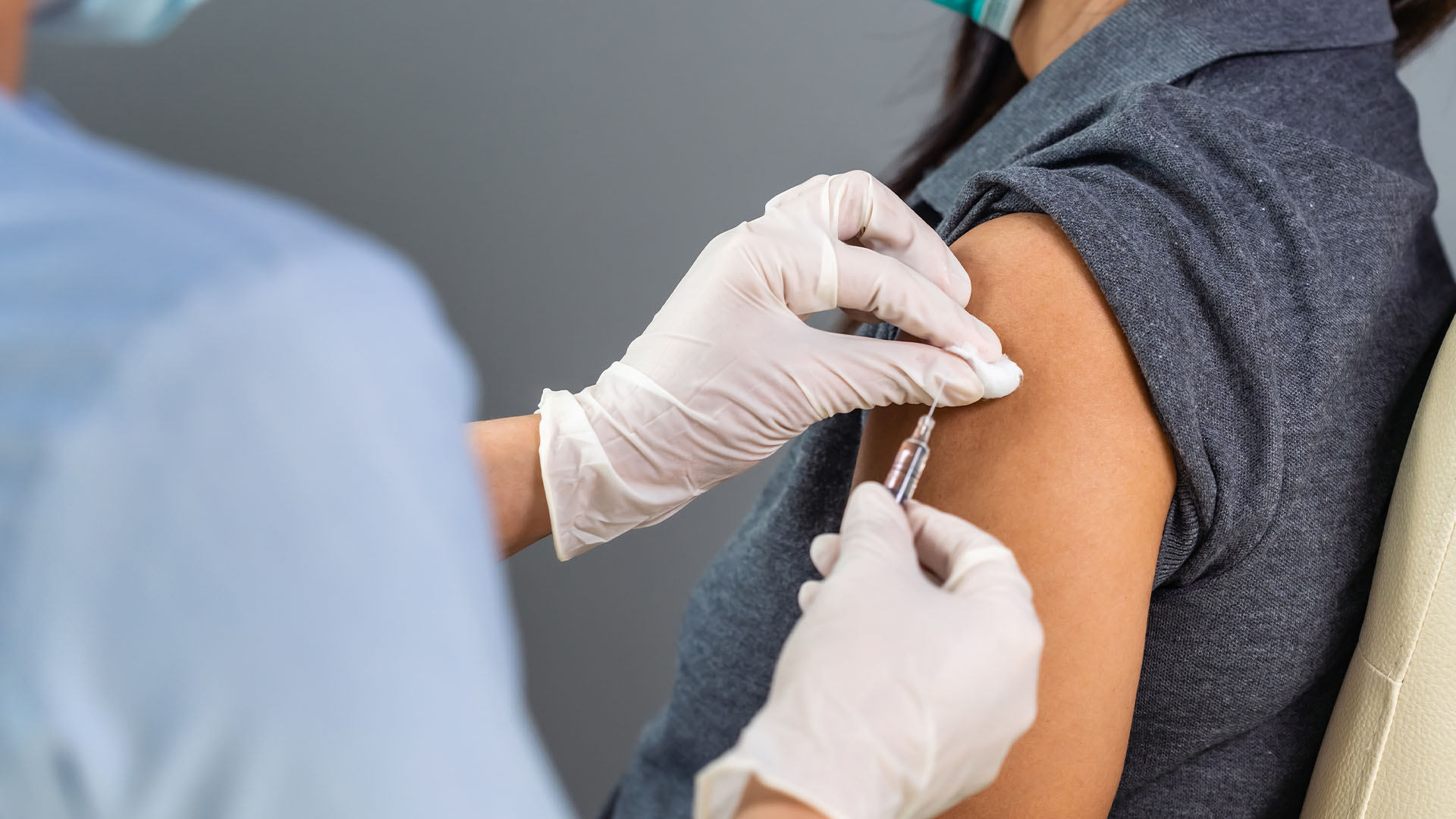
The No-Fault Scheme commenced on 6 September 2021 and it has been backdated to cover any adverse reactions to vaccinations from February 2021. The Scheme applies to anyone who receives a COVID-19 vaccine from any of the providers linked to the government-approved COVID-19 vaccination program. The Scheme will cover the costs of injuries above $5,000 due to a proven adverse reaction to a COVID-19 vaccination.
Patients wishing to seek compensation between $5,000 and $20,000 will need to express interest in making a claim, and provide applicable evidence of hospitalisation, the nature of the injury, medical costs and lost wages. Evidence requirements for patients seeking $20,000 and above are still being developed. That claim will be assessed by independent experts who will consider liability and costs. The Scheme is to be fully funded by the Australian Government and compensation paid based on the recommendations of independent experts.
Precise details of the operation of the Scheme are still emerging, however, it appears that the Scheme will essentially be a reimbursement programme for patients suffering an adverse reaction to recoup treatment expenses as well as lost wages. The precise definition of “adverse reaction” to be applied is not yet clear, including whether it is restricted to reaction to the medication itself (as opposed to say, infections from needle-stick injuries). However, it is anticipated that the Australian Government will not adopt a restrictive approach to eligibility under the Scheme – for example limiting eligibility for compensation to those who meet a certain percentage of disablement as applied in the UK Vaccine Damage Payment Scheme.
While unclear, it is unlikely that the Scheme will prevent patients seeking compensation through traditional legal paths where fault or legal liability of the manufacturer must be established. Such litigation seems possible noting that the Scheme does not cover the larger damages components usually recovered in product liability actions – in particular damages for pain and suffering, future income loss, and costs of care and assistance.
How the government’s indemnities to manufacturer’s will interplay with such actions is unclear given the precise scope of the indemnities has not yet been made public. However, it would appear most likely that any action would still be brought against vaccine suppliers, manufacturers, and healthcare professionals in the same way as traditional pharmaceutical product and healthcare liability lawsuits – that is, individual or class actions based on breaches of the Australian Consumer Law and common law duties of care. It is likely that upon receipt of a claim, the manufacturer would need to approach the Australian Government to recover under the indemnity.
It also remains to be seen if the unprecedented circumstances of the development, regulatory approval and supply of Covid vaccines will provide additional defences to vaccine manufacturers, healthcare professionals and the Australian Government, beyond the usual defences of what a manufacturer must reasonably do before bringing a medicine to market, or what a healthcare professional must do before prescribing and administering it. The reasonableness of supply, prescription and administration in such circumstances is, not surprisingly, untested. However, it is something the courts may be grappling with regularly in the coming years.

Publication
On August 1, 2025, the UK Supreme Court delivered its long-awaited judgment in Hopcraft v Close Brothers Limited and on 3 August the FCA announced it would consult on a redress scheme.
Publication
The Regional Court of Munich (LG München I) has issued a landmark judgment in GEMA v OpenAI (Case No. 42 O 14139/24), holding that the use of copyrighted song lyrics for training generative AI models without a licence violates German copyright law.
Publication
Songa Product and Chemical Tankers III AS v Kairos Shipping II LLC [2025] EWCA Civ 1227 (07 October 2025) has clarified the extent of the obligation on the Charterer to redeliver a vessel following the termination of a Barecon 2001 charter and of the Owner’s right to require it to be redelivered to a port “convenient to them”.
Subscribe and stay up to date with the latest legal news, information and events . . .
© Norton Rose Fulbright LLP 2025