
Publication
Federal budget 2025: Updates for federally regulated employers
The federal government’s recently released Canada Strong Budget 2025 (Budget 2025) has proposed several measures with implications for federally regulated employers.


Australia | Publication | July 8, 2019
Conditions attached to the licensing or assignment of intellectual property (IP) have benefited from an exception to certain provisions of the Competition and Consumer Act 2010 (Cth) (CCA) under section 51(3) (IP Exception). The IP Exception previously excused conduct even if it was anti-competitive conduct, in purpose or effect, or cartel conduct, provided it was not a misuse of market power or resale price maintenance by reason of IP licensing or assignment.
From 12 September 2019, any arrangements that have relied on the long-standing exception will now be subject to all prohibitions, including cartel conduct, and under scrutiny as to the purpose or the effect of the restrictions attached to the granting of IP rights. The repeal of the IP Exception is particularly significant for the pharmaceutical and life sciences sector. Where businesses in this sector regularly license or assign conditionally their IP rights as a way of protecting and monetising their R&D investment and also as part of settling IP disputes, there are clearly identifiable points of exposure. Indeed, one of the drivers for the repeal was the perception that in sectors with multiple and competing IP rights, such as the pharmaceutical sector, cross-licensing arrangements may be entered into to resolve disputes which may impose anti-competitive restrictions on each licensee.
Any licensing agreement that potentially affects competition, even if the agreement has been in place for some time, should be reviewed now to ensure competition compliance going forward. The CCA carries significant consequences including hefty penalties.
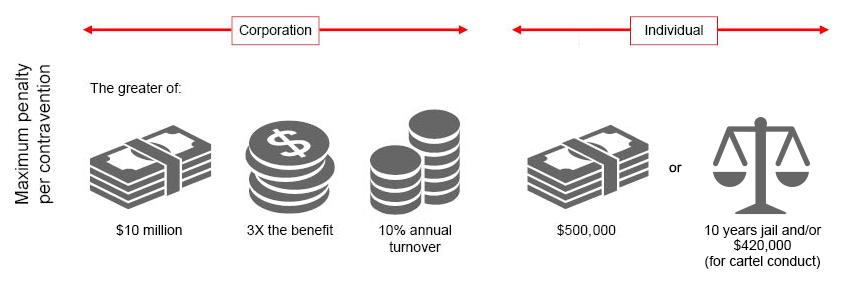
Section 51(3) of the CCA currently provides that certain conditions imposed in licences or assignments of IP rights, including patents, registered designs and copyright, are excluded from certain anti-competitive conduct, such as cartel conduct and exclusive dealing. Consequently, IP owners have been in a position to impose restrictions on the use of their IP that may be anti-competitive.
Removal of the IP Exception will expose businesses that have relied on it to the full force of the CCA. Stipulation of the conditions under which a licensee can use licensed IP has been a powerful and often used approach by which an IP owner controls the use of their IP assets in whatever manner they see fit. Going forward, the purpose and the effect of those conditions will need to be examined through a competition lens. This will be particularly important where the relevant agreements are between competitors.
Importantly, there will be no “grandfathering” of the current exception – the removal of the IP Exception will apply to new and pre-existing arrangements in place from the date the change comes into effect. This means that current agreements, irrespective of when they were entered into will be subject to the Act.
Businesses in the pharmaceutical and life sciences sector will be most affected by the repeal when relying on the ownership and licensing of intellectual property rights to protect and monetise their R&D investment. There are a number of readily identifiable key risk areas:
|
Patent Pools |
These are agreements between patent owners to license one or more of their patents to one another or to third parties. This poses a risk of undermining competition, or potentially reducing innovation incentives. |
|
Cross-licensing |
Cross-licensing involves an agreement between two parties to grant rights to their IP rights to the other. This runs the risk of breaching the cartel conduct provisions under the CCA or risk falling foul of the provisions which prohibit agreements which may substantially lessen competition. |
|
Conditional licensing |
For example:
|
|
Licence term |
Licences, where the term extends beyond the life of the licensed IP. |
|
Potential licensee challenges |
Potential for licensees challenging the terms of existing licensing arrangements where they may be contrary to the new law. In particular, the inclusions of conditions which restrict the ability of the licensee to compete with the IP owner or restrict the licensee in terms of pricing, territory or customers where this may be anti-competitive. |
|
IP enforcement and settlement agreements |
IP enforcement and settlement strategies typically involve consideration of who to enforce against or settle with and on what terms. Such strategies will now need to be analysed through a widened competition lens, with the rationale for the action particularly in focus. |
|
“Pay-for-delay” agreements |
These agreements are used to settle patent infringement litigation, where the patent owner pays the alleged infringer to settle the dispute. In the pharmaceutical context, such agreements typically involve the patent owner paying the generic manufacturer not to enter the market in circumstances where the generic may not infringe the patent or where the agreed entry date is significantly later than the date on which the patent expires. The restrictions on use of patents inherent in these agreements pose a risk of misuse of market power by the IP rights holder (unchanged by the IP Exception) and call into question the anti-competitive purpose of any arrangement. |
|
Market-specific concerns |
Competition concerns are more likely to arise in markets where there is low substitutability, for example, where a patented drug is the standard of care. |
A misuse of the market power or resale price maintenance conduct were excluded from the application of the exception.
Certain other exceptions may apply to cartel conduct under the CCA, such exceptions include matters relating to remuneration or conditions of employment, conduct subject to a collective bargaining notifications, certain joint ventures, exclusive dealing and agreements between related companies.
The “purpose” of the conduct or course of action is critical. An overarching commercial purpose must be paramount. In considering the “effect” of any arrangement, it is important to note that the existence of a patent may give rise to a single product market. This effectively limits competition and heightens the risk that conduct may raise competition concerns.21
On 21 June 2019, the ACCC published draft guidelines on the application of competition laws to IP rights following the removal of the exception. The ACCC seeks the public’s view on its draft guidelines, with submissions able to be lodged with the ACCC until 19 July 2019. As currently drafted, these guidelines set out the general principles that will guide the ACCC’s approach to compliance and enforcement of the competition laws to conduct involving intellectual property rights; outlines the types of conduct that are or will now be subject to the competition laws; and provides examples to illustrate conduct that the ACCC considers is likely to contravene the competition laws.
Immunity from the application of competition laws to IP arrangements can be sought where the arrangement is likely to result in a net benefit to the public, by notifying or applying for authorisation from the ACCC.
All businesses with existing IP licensing or assignment arrangements should review their agreements to ensure they comply with the prohibitions against anti-competitive conduct under Part IV of the CCA.
Generally, any arrangements that provide for territorial licence restrictions, conditions attaching to the use of IP rights for particular customers or groups of people, quality restrictions, or price will need to be considered for compliance with the Act.
Agreements between competitors must ensure that the IP licensing / assignment agreements do not run the risk of:
Preparing a list of all agreements and considering the type of licence and the implications of the arrangements is the key starting point. Where required, agreements may need to be re-negotiated to ensure compliance going forward.
The Treasury Laws Amendment (2018 Measures No. 5) Bill 2018 was given Royal Assent on 12 March 2019 (now the Act), after passing both Houses of the Australian Government in mid-February. It will enact recommendations from both the Harper Review and Productivity Commission’s IP Arrangements Inquiry by repealing s 51(3) of the CCA.
The Harper Review came to the conclusion that, whilst in the majority of cases granting an IP right is unlikely to raise significant competition concerns, IP rights, like all property rights, can be used in a manner that harms competition. The Harper Review recommended that arrangements in relation to IP should be examinable under the competition law. Adding to this, the Productivity Commission argued that commercial transactions involving IP rights should be subject to the CCA in the same manner as transactions involving other property rights and assets. The Productivity Commission also considered that the benefits of removing the IP Exception could rise in the future as the level of licensing and cross licensing increases, especially in the pharmaceutical sector.
Removing the IP Exception will also bring Australia in line with other comparable international jurisdictions, such as the US, Canada and Europe, which do not have an IP Exception in competition laws. Recent areas of focus for regulators in the US, Canada and Europe have been on the anti-competitive effects of ‘pay for delay’ arrangements in patent settlements, in particular arrangements which may deter entry by generic pharmaceutical manufacturers or other cases involving an IP owner abusing a dominant position (e.g. recent actions by the European Commission involving Google and Qualcomm).
Businesses have until 12 September 2019 to review any licensing agreements or agreements that provide for the licensing of IP to ensure compliance with the Act. Agreements between competitors (that are not subject to any other exception) are particularly high risk, and the purpose of the restraint, in addition to the effect of the arrangement, must be considered to ensure that the arrangements are not anti-competitive.
Please contact us for further information.

Publication
The federal government’s recently released Canada Strong Budget 2025 (Budget 2025) has proposed several measures with implications for federally regulated employers.

Publication
On 13 November 2025, the Financial Reporting Council (FRC) published its Annual Review of Corporate Governance Reporting. This analyses reporting trends and practices among 100 UK-listed companies against the 2018 UK Corporate Governance Code (Code).
Publication
On 13 November 2025, the European Parliament adopted (subject to certain amendments) the substantive Omnibus Directive which was proposed by the European Commission on 26 February 2025 (see our previous briefing here). The Omnibus proposal has now been referred to the Committee of Legal Affairs to proceed to the trilogue negotiations.
Subscribe and stay up to date with the latest legal news, information and events . . .
© Norton Rose Fulbright LLP 2025