In a January 23, 2019 precedential opinion, the Federal Circuit determined that the US Patent and Trademark Office (USPTO) has been incorrectly determining the length of an additional patent term for which a patentee may qualify. In Supernus Pharm., Inc. v. Iancu, the Federal Circuit panel reversed the US District Court of the Eastern District of Virginia’s grant of summary judgment to the USPTO, finding that the USPTO had incorrectly reduced patent term adjustment (PTA) for applicant delay when, under the circumstances presented, the applicant could not have made any identifiable efforts to advance prosecution during that time period. No. 2017-1357 (Fed. Cir. Jan. 23, 2019). The Federal Circuit held that the USPTO’s calculation of PTA exceeded its authority under the statute governing PTA.1 See 35 U.S.C. § 154. The court held that the USPTO is statutorily authorized to deduct PTA for applicant delay only when the applicant failed to engage in reasonable efforts to conclude prosecution, and remanded the case for further proceedings. Here, there were no reasonable efforts the applicant could have taken, and, therefore, the USPTO incorrectly deducted PTA for that time period.
Supernus Pharmaceuticals, Inc. is the owner and assignee of US Patent No. 8,747,897 (the ’897 patent),2 and United Therapeutics Corp. is the exclusive licensee of the ’897 patent, (collectively, the owner-assignee and the licensee will be referred to as Supernus). A series of events occurred during the prosecution of US Patent Application No. 11/412,100 (the ’100 application; later issuing as the ’897 patent) that were taken into consideration in deciding the amount of applicant delay that should be deducted from the amount of USPTO delay in calculating PTA. The relevant events began when the USPTO issued a final rejection on August 20, 2010. On February 22, 2011, Supernus filed a request for continued examination (RCE). On September 11, 2012, Supernus was notified by its European counsel that the European Patent Office (EPO) had informed European counsel on August 21, 2012, of a competitor’s filing of a Notice of Opposition on a European patent that claimed priority to the ’100 application. Supernus then filed a supplemental information disclosure statement (IDS) with the USPTO on November 29, 2012 (100 days post-EPO notification of the filing of the opposition), disclosing the European opposition and providing the USPTO with the EPO notification, the opposition documents, the documents cited within the opposition, and the letter from its European patent counsel.
Upon issuance of the ’897 patent, the USPTO calculated 1,260 days of PTA, with 2,321 days attributed to USPTO delays3 (1,656 days for Type A delays and 665 days for Type B delays, and reduced by 175 days of overlapping Type A and Type B delays) and 886 days attributed to applicant delay. Of the 886 days calculated for applicant delay, 646 days were attributed to the period between the filing of the RCE and the filing of the supplemental IDS.
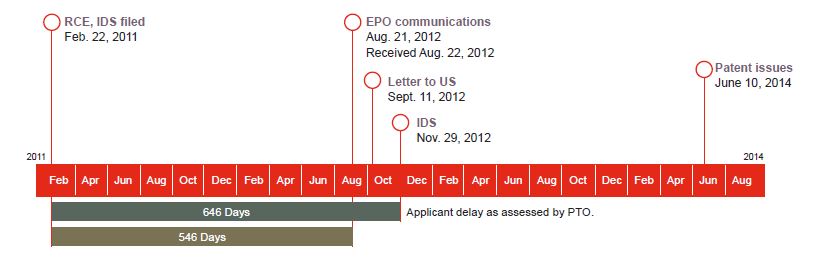
According to the USPTO, Supernus’ filing of the supplemental IDS after a RCE had been filed, absent an express request by the examiner, constituted applicant delay pursuant to USPTO regulations under 37 C.F.R. § 1.704(c)(8).4 Supernus filed a request for Reconsideration of Patent Term Adjustment arguing that the 646-day PTA deduction under 37 C.F.R. § 1.704(c) (8) was improper. The USPTO rejected Supernus’ request for reconsideration and Supernus appealed to the district court. The district court granted the USPTO summary judgment, as a matter of law, based on Gilead Scis., Inc. v. Lee, 778 F.3d 1341 (Fed. Cir. 2015). Supernus Pharm., Inc. v. Lee, No. 1:16- CV-00342, 2016 WL 8739331 (E.D. Va. Oct. 18, 2016). In Gilead, the Federal Circuit upheld a PTA reduction under 37 C.F.R. § 1.704(c)(8) where the applicant filed a supplemental IDS disclosing other co-pending patent applications after its response to a restriction requirement. 778 F.3d 1341. The district court stated that 37 C.F.R. § 1.704(c)(8) “applie[d] squarely to this case” and that the Federal Circuit’s decision in Gilead foreclosed Supernus’ claim against 37 C.F.R. § 1.704(c) (8). Supernus, 2016 WL 8739331, at *11.
On appeal, the Federal Circuit held that the district court erred in finding that Gilead foreclosed Supernus’ statutory interpretation argument. The court stated that Gilead addressed “whether a failure to engage in reasonable efforts requires conduct that actually causes delay.” Supernus, slip op. at 12. Here, Supernus’ argument concerned whether the reduction in PTA can exceed the “‘time during which the applicant failed to engage in reasonable efforts to conclude prosecution of the application.’ 35 U.S.C. § 154(b)(2)(C)(i).” Id. at 11. The Federal Circuit noted that in Gilead, the patent owner could have taken action and filed the supplemental IDS when it filed its response to the restriction requirement, instead of filing the supplemental IDS later. Id. at 12. In contrast, “Supernus could not have undertaken any efforts to conclude prosecution of the ’100 application” in the 546-day period between the filing of the RCE and the EPO’s notification to Supernus’ European counsel of the filing of an opposition. Id.
Applying the Chevron framework for statutory interpretation,5 the Federal Circuit concluded that the plain language of the PTA statute clearly indicated “that the USPTO may not count as applicant delay a period of time during which there was no action that the applicant could take to conclude prosecution of the patent. Doing so would exceed the time during which the applicant failed to engage in reasonable efforts.” Id. at 14. As the court noted, the PTA statute itself states: “The period of adjustment of the term of a patent under paragraph (1) shall be reduced by a period equal to the period of time during which the applicant failed to engage in reasonable efforts to conclude prosecution of the application. 35 U.S.C. § 154(b)(2)(C)(i) (emphases added).” Id. Thus, the USPTO could not count as applicant delay the time period in which no identifiable efforts could have been undertaken by Supernus, i.e., the 546-day period6 after Supernus filed the RCE and before it was notified by the EPO of the filing of the opposition. Id. at 17. The Federal Circuit concluded that the USPTO’s application of its PTA regulations to be inconsistent with the PTA statute and, thus, no deference was due to the USPTO’s interpretation.
As a result, for recently issued patents, patent owners with similar circumstances as Supernus may potentially be able to reclaim PTA that had been reduced as a result of a supplemental filing after a reply. However, patent owners will still have to demonstrate that “reasonable efforts” had
been undertaken by the applicant during that time period. Also, patent owners must act quickly to review the USPTO’s calculations of PTA and timely petition for a request for reconsideration of PTA. Patent owners have two months (extendible by up to an additional five months7) after the date the patent was granted to file a request for reconsideration of PTA. 37 C.F.R. § 1.705. If the USPTO has denied a request for reconsideration of PTA, the applicant can then file an appeal in the US District Court for the Eastern District of Virginia within 180 days of the USPTO’s decision. 35 U.S.C. § 154(b)(4).





